
The Javelin Platform

Where human biology drives preclinical confidence.
The Javelin Platform recapitulates human physiology with a high-fidelity microphysiological system, capturing critical drug and disease responses that traditional models miss to provide clinically predictive insights across therapeutic modalities.
DESIGNED FOR SUCCESS
A liver-centric, multi-organ system engineered to support native tissue-like MPS culture with primary human cells - giving you a physiologically relevant window into human drug response.
A next-generation bioengineered platform with a user-centric design that requires minimal training and integrates seamlessly into your existing preclinical workflow, delivering all the power of human MPS without the operational burden.
An assay-agnostic platform that provides the ability to develop novel assays or adapt existing biochemical, imaging-based, or omics-driven protocols without platform-imposed boundaries. Unparalleled flexibility of detection method, sampling strategy, and analytical pipeline in a translational human MPS system.
LIVER-CENTRIC ARCHITECTURE
Physiologically Relevant MPS
Human primary cells in a spatially organized architecture recapitulates the structural and functional complexity of human liver
Transcriptomic fidelity to human liver with sustained receptor expression enables extended evaluation of advanced modalities
Vascular-first delivery via recirculating flow mimics systemic exposure and in vivo-like sequential drug interactions
Clinically Relevant Drug Exposure
Validated for 21+ day repeat-dose studies with flexible dosing schedule comparable to clinical regimens
Facilitates assessment of cumulative toxicity and long-term therapeutic effects under clinically and physiologically relevant dose and exposure conditions
Extended culture stability overcomes lifespan limitations and media change dependency of competing platforms
High-Content, Human-Relevant Data
Generate rich datasets that correlate more closely with clinical outcomes than conventional preclinical models.
Multi-parametric assessment of clinical biomarkers from single study emphasizing efficiency and mechanistic depth.
Comprehensive preclinical profiling from a consistent experimental system, from PKPD and hepatotoxicity to multi-organ liability assessment
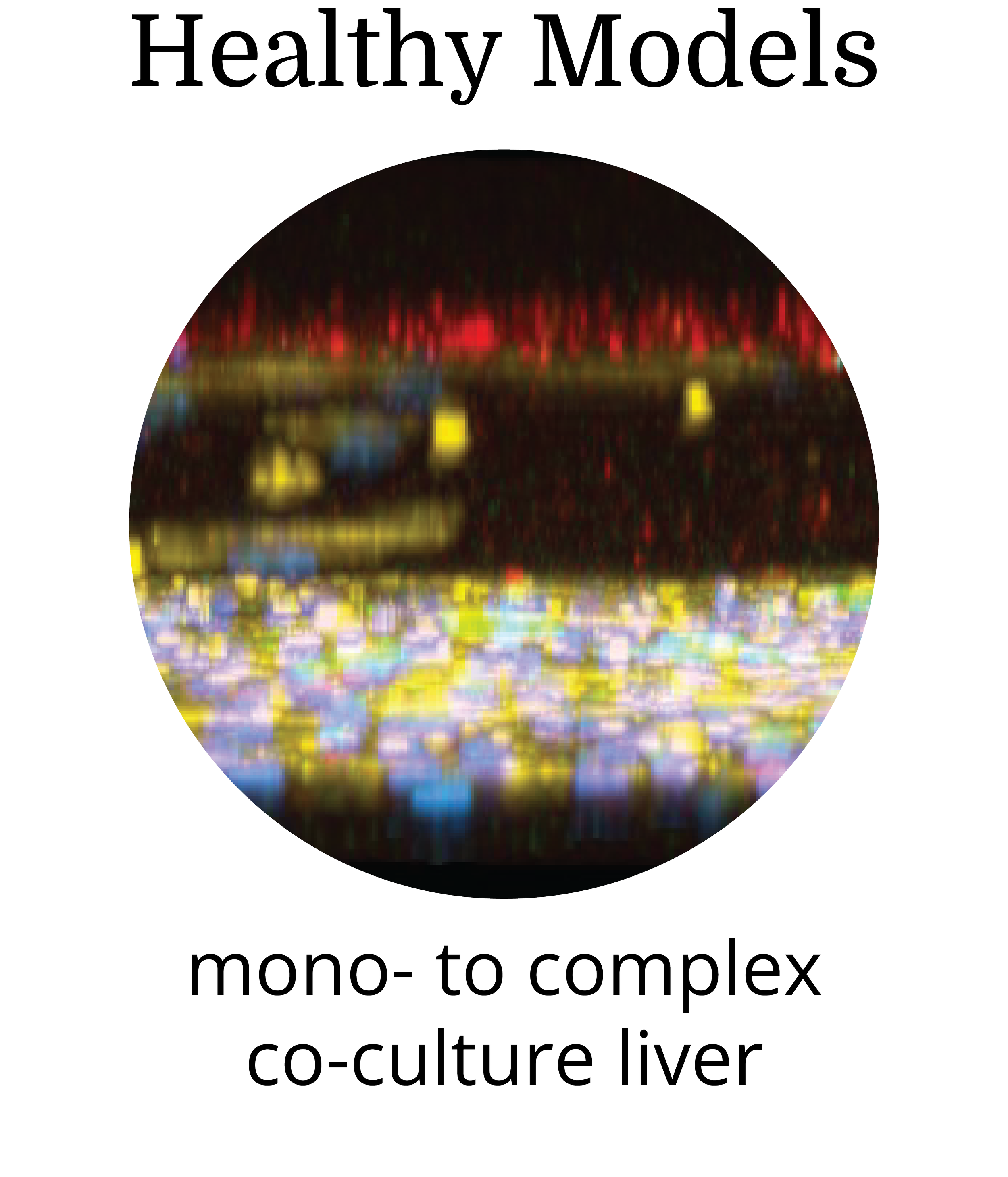

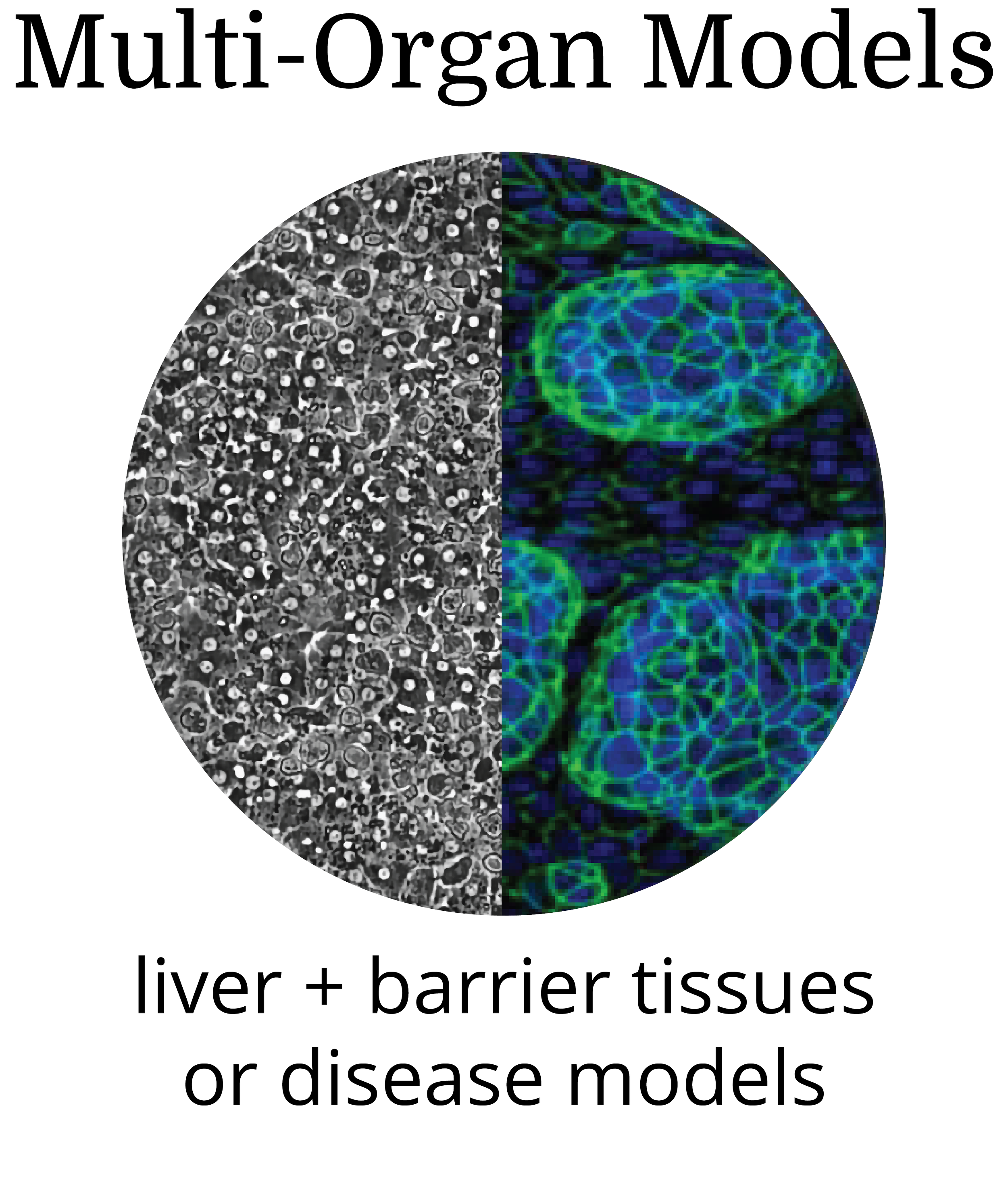

An in vitro New Approach Method (NAM) that delivers clinically actionable and predictive insights
Clinically translatable
Reduced animal use
3Rs compliant
Advance your pipeline with confidence.
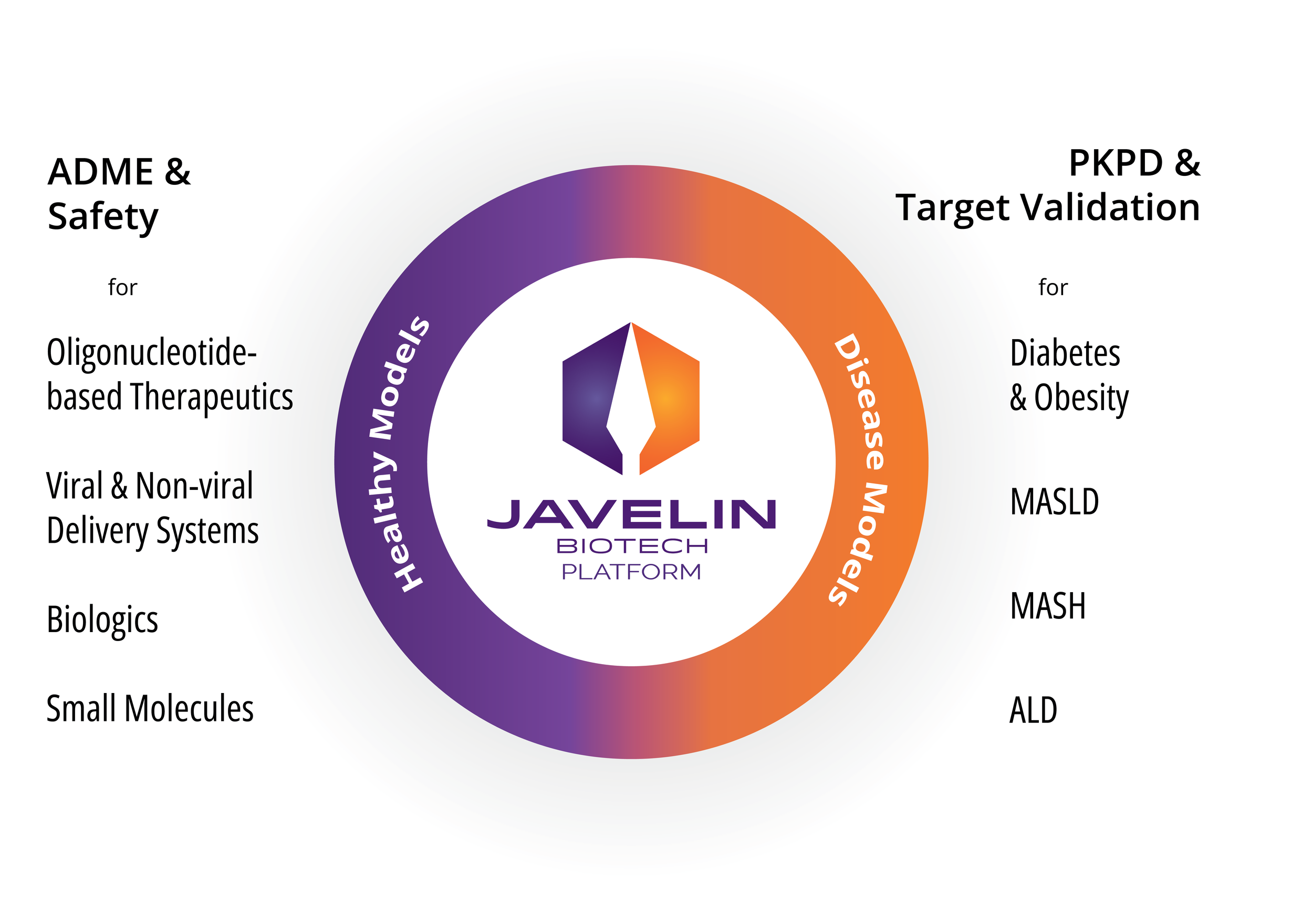
APPLICATIONS ACROSS PRE-CLINICAL DISCOVERY
Reduce your reliance on animal studies during lead optimization with a NAM that delivers the human-relevant mechanistic insights regulators and sponsors need.
Compliment and reduce animal studies
The Javelin Platform is purpose-built as a NAM for lead optimization and preclinical assessment – reducing and refining animal use while improving translational fidelity.
Capture what animal models miss
Complex multi-organ drug interactions, multiple modes of hepatotoxicity, and species-specific metabolism gaps are addressed with primary human cells in a physiological context.
Supports traditional and advanced therapeutics
From small molecules to cell and gene therapies, the Javelin Platform provides mechanistically rich data across diverse modalities.
Regulatory-aligned strategy
Positioned for the FDA Modernization Act 2.0 & 3.0 era, generate NAM data that supports IND packages and reduces late-stage attrition risk.
